Greenhouse effect
Some heat energy from the Earth’s surface escapes into space. If too much heat energy escaped, the planet would be very cold. However some gases in the atmosphere can trap escaping heat energy, causing some of it to pass back to the surface.
These are called greenhouse gases, and they keep our planet warm, which is a good thing. Carbon dioxide is an important greenhouse gas.
Increasing carbon dioxide levels
Humans burn fossil fuels such as oil, coal and natural gas. The energy released is used to power cars and other machines, to generate electricity, and to keep buildings warm. The burning fuel releases waste gases, including carbon dioxide. As the human population increases, more fuel is used, and more carbon dioxide is released.
Global warming
The extra carbon dioxide increases the greenhouse effect. More heat is trapped by the atmosphere, causing the planet to become warmer than it would be naturally. The increase in global temperature this causes is called global warming.
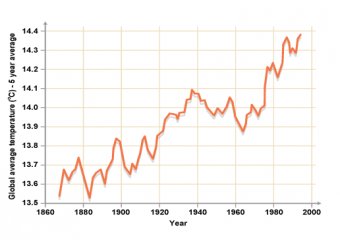
The amount of carbon dioxide in the air has increased a lot in recent years
The average temperature of the planet has increased in recent years
Climate change
Global warming is beginning to cause big changes in the environment. These include:
- ice melting faster than it can be replaced in the Arctic and Antarctic
- the oceans warming up – their water is expanding and causing sea levels to rise
- changes in where different species of plants and animals can live
The extra heat energy is also beginning to cause large-scale changes in the weather. These changes in weather patterns around the world are called climate change.
RELATED VIDEO